What is pH?
Table of contents
[MISSING: System.API.no-headers]pH is a measurement that shows the ratio of +/- hydrogen ions in concentration and is commonly used in the analysis of soil and water testing. The concept of pH was first introduced by Soren Sorensen in 1909 and revised to the modern pH scale in 1924 which is measured on a scale of 0 to 14 shown below.
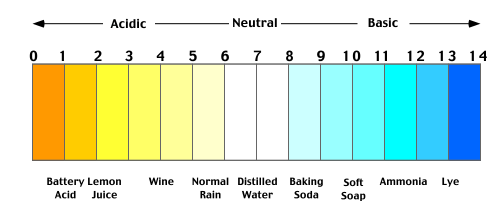
In general terms pH of 7 is neutral, pH less than 7 is acidic, and pH greater than 7 is basic, therefore battery acid is more acidic than ammonia and baking soda is more basic than wine. The modern pH scale is logarithmic, which means that a unit decrease in pH is equal to a ten-fold increase in acidity e.g. rain (approx. pH 6) is at least ten times more acidic than distilled water (approx. pH 7).
Most fish will happily live in a wide range of pH’s but are slow to adapt to a change in pH. Therefore a sudden change in pH can be very dangerous to fish, plants, bacteria and other living creatures. A seneye device alerts you to sudden changes in the aquarium.
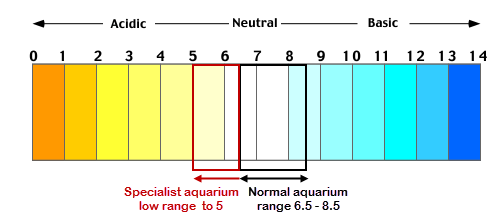
As a rule of thumb pH change in the aquarium should be minimised, anything above pH of 0.5 per day is counted as deadly or bad. Often aquarists changing water chemistry to reach absolute perfect values will cause more harm than good. If the fish are happy then a controlled and constant pH is most important. It is not uncommon to measure a big change when any pH device is recalibrated or new reagents are used; this does not mean the water has changed.
Daily swings in pH are common in the aquarium; the size of the swing is usually controlled by the hardness of the water (KH). This means marine systems will see very small swings as they have high buffer capacities (KH), however soft freshwater aquariums (especially Amazonian or black water tanks) will not normally have high KH and will react greater.
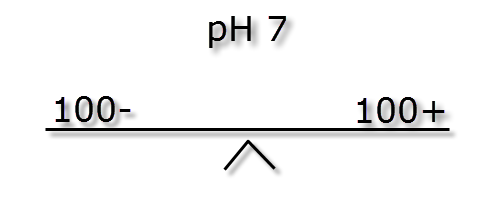
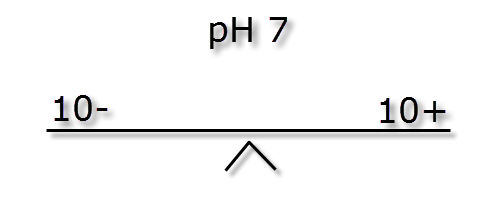
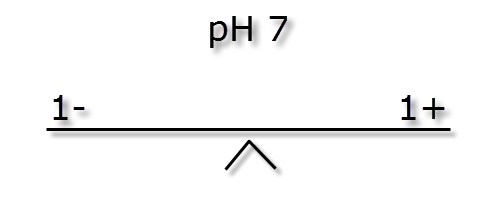
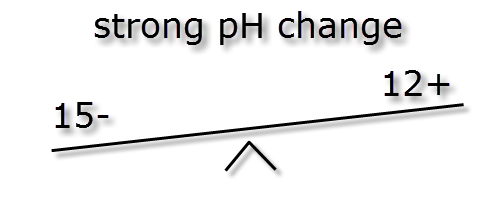
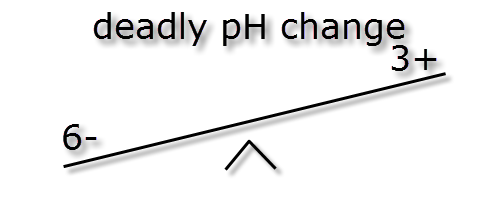
In the following passage we will try to explain how pH and KH are linked. As mentioned earlier the pH measurement is a ratio, how easy it is to change this ratio (pH stability) is dictated by the ionic (buffer) strength also known as KH. The images below illustrate how different ionic strengths can report the same pH value. If you imagine equal + hydrogen ions on one side of a balance to – hydrogen ions the other side then the level will flat or neutral. This would represent a pH of 7 and the absolute number of + or – ions on each side is not important so long as they balance.
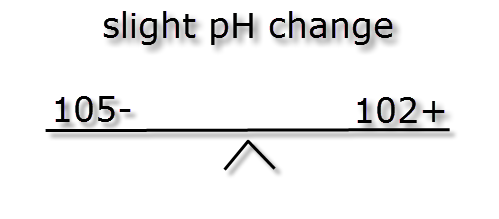
If we were to mix the three different buffer strength waters shown above with and equal volume of water of a low pH (acidic) and weak buffer strength, say -5 ions & + 2 ions we can see the effect below.
As you can see the more highly buffered water doesn’t move much but the low buffered water moves huge amounts. Therefore the higher the buffering KH the more stable the pH. This is also why using RO water (which has no buffer strength) to lower pH will not work, it will however reduce the KH and pH stability.
A low KH will also make and pH changes hard as pH will usually revert back to where it was; the ability to resist pH change is often called alkalinity. Eventually a low KH will also often lead to pH crashes.
If you have a problem and do need to adjust pH up or down we recommend using both acid and base buffers; and always make changes over a long period.
In the fresh water aquarium is plants which use CO2 present in the water throughout the day during a process called photosynthesis. CO2 is a gas dissolved in the aquarium water and that makes it more acidic (like fizzy drinks), when plants have no light CO2 starts to build up and lower pH.
Not getting the pH results you expected then work through our check list and Click here to learn more...
SENEYE USERS WITH LOW KH (SOFTWATER) TANKS CLICK HERE